Fearless Energy Drink Potentiates Anxiety, Oxidative Stress, and Neuronal Cell Damage on the Hippocampus of Adult Male Wistar Rat
DOI:
https://doi.org/10.60787/tnhj.v23i4.746Keywords:
Neuroscience, Hippocampus, Fearless, Energy drinks, Anxiety, Oxidative stressAbstract
Background: The relationship between the impact of Fearless energy drinks on the hippocampus has not yet been documented. Study investigated the effect of Fearless energy drinks on oxidative stress and hippocampal microstructure in Wistar rats.
Method: Twenty male rats weighing 160-200g were divided into four groups of five rats each. Group A was the control, groups B, C and D received Fearless energy drink orally using gastric tube with the following doses: 2.5ml, 5ml and 7.5ml/kg/day respectively for three weeks. At the end, elevated plus maze test was carried out to check for anxiety. The animals were sacrificed and oxidative stress markers such as superoxide dismutase (SOD) and malondialdehyde (MDA) were carried out; the brains were dissected for hippocampal specimens, which were processed for histological studies using hematoxylin and eosin method.
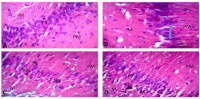
Result: There was an increased level of anxiety-like behavour in animals in the experimental groups, as they spent more time in the close arm of the maze than in the open arm of the maze when compared to the control. There was a significant decrease (p ≤ 0.05) in SOD and a significant increase (p ≤ 0.05) in MDA, which indicate oxidative stress. Histological examination of the hippocampus from Fearless EDs groups showed vacuolation, hypertrophied pyramidal cells with numerous pyknotic nuclei, cracks in pyramidal cell layer and decreased cellular density in the pyramidal cell layer when compared to the control.
Conclusion: Fearless energy drinks administration induced anxiety, deficient antioxidant capacity and morphological change on the hippocampus of Wistar rats.
Downloads
References
Iyaniwura O, Abdulfatai A. Toxicodynamic Effects of ‘Red Bull’ Energy Drink in A Randomised Controlled Study on Local Strains of Adult Rabbits. 2018;9(1):46–64.
Seifert SM, Schaechter JL, Hershorin ER, Lipshultz SE. Health effects of energy drinks on children, adolescents, and young adults. Pediatrics. 2011;127(3):511–28.
Wolk BJ, Ganetsky M, Babu KM. Toxicity of energy drinks. Curr Opin Pediatr. 2012;24(2):243–51.
Glade MJ. Caffeine-Not just a stimulant. Nutrition. 2010;26(10):932–8.
Smith A. Effects of caffeine on human behavior. Food Chem Toxicol an Int J Publ Br Ind Biol Res Assoc. 2002;40(9):1243–55.
Prins PJ, Goss FL, Nagle EF, Beals K, Robertson RJ, Lovalekar MT, et al. Energy Drinks Improve Five-Kilometer Running Performance in Recreational Endurance Runners. J strength Cond Res. 2016;30(11):2979–90.
Costa-Valle MT, Gomes JF, De Oliveira CR, Scherer A, Franco De Oliveira SCW de SE, Menezes RCR, et al. Energy drinks and alcohol in a binge drinking protocol in Wistar rats: Male and female behavioral and reproductive effects. Pharmacol Biochem Behav [Internet]. 2022; 221:173487. Available from: https://www.sciencedirect.com/science/article/pii/S0091305722001666
Dahl RE. Adolescent brain development: a period of vulnerabilities and opportunities. Keynote address. Ann N Y Acad Sci. 2004; 1021:1–22.
Campbell B, Wilborn C, La Bounty P, Taylor L, Nelson MT, Greenwood M, et al. International Society of Sports Nutrition position stand: energy drinks. J Int Soc Sports Nutr. 2013;10(1):1.
Grasser EK, Miles-Chan JL, Charrière N, Loonam CR, Dulloo AG, Montani JP. Energy Drinks and Their Impact on the Cardiovascular System: Potential Mechanisms. Adv Nutr. 2016;7(5):950–60.
Attila S, Çakir B. Energy-drink consumption in college students and associated factors. Nutrition. 2011;27(3):316–22.
Guilbeau JR. Health risks of energy drinks: what nurses and consumers need to know. Nurs Womens Health. 2012;16(5):423–8.
Tibrewal P, Dhillon R. Caffeine induced psychotic exacerbation. Vol. 45, The Australian and New Zealand Journal of Psychiatry. 2011; 45(2): 179 – 180.
Ishak WW, Ugochukwu C, Bagot K, Khalili D, Zaky C. Energy drinks: psychological effects and impact on well-being and quality of life-a literature review. Innov Clin Neurosci. 2012;9(1):25–34.
Bliss T. The hippocampus book. Andersen P, Morris R, Amaral D, O’Keefe J, editors. The hippocampus book. New York, NY, US: Oxford University Press; 2007. xx, 832–xx, 832.
Liu Y, Guan W, Ren G, Yang Z. The possible mechanism of silver nanoparticle impact on hippocampal synaptic plasticity and spatial cognition in rats. Toxicol Lett. 2012;209(3):227–31.
Siddiqui SV, Chatterjee U, Kumar D, Siddiqui A, Goyal N. Neuropsychology of prefrontal cortex. Indian J Psychiatry. 2008;50(3):202–8.
El Falougy H, Kubikova E, Benuska J. The microscopical structure of the hippocampus in the rat. Bratisl Lek Listy. 2008;109(3):106–10.
Huang TT, Zou Y, Corniola R. Oxidative stress and adult neurogenesis--effects of radiation and superoxide dismutase deficiency. Semin Cell Dev Biol. 2012;23(7):738–44.
Chang BJ, Jang BJ, Son TG, Cho IH, Quan FS, Choe NH, et al. Ascorbic acid ameliorates oxidative damage induced by maternal low-level lead exposure in the hippocampus of rat pups during gestation and lactation. Food Chem Toxicol an Int J Publ Br Ind Biol Res Assoc. 2012;50(2):104–8.
Licht T, Kreisel T, Biala Y, Mohan S, Yaari Y, Anisimov A, et al. Age-Dependent Remarkable Regenerative Potential of the Dentate Gyrus Provided by Intrinsic Stem Cells. J Neurosci Off J Soc. Neurosci. 2020;40(5):974–95.
Díaz A, Treviño S, Guevara J, Muñoz-Arenas G, Brambila E, Espinosa B, et al. Energy Drink Administration in Combination with Alcohol Causes an Inflammatory Response and Oxidative Stress in the Hippocampus and Temporal Cortex of Rats. Oxid Med Cell Longev. 2016; 2016:8725354.
Al-Basher GI, Aljabal H, Almeer RS, Allam AA, Mahmoud AM. Perinatal exposure to energy drink induces oxidative damage in the liver, kidney and brain, and behavioral alterations in mice offspring. Biomed Pharmacother. 2018; 102:798–811.
Halliwell B. Free radicals, proteins and DNA: oxidative damage versus redox regulation. Biochem Soc Trans. 1996;24(4):1023–7.
Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M, Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39(1):44–84.
Olanow CW. An introduction to the free radical hypothesis in Parkinson’s disease. Ann Neurol. 1992;32 Suppl: S2-9.
Walf AA, Frye CA. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat Protoc [Internet]. 2007;2(2):322–8. Available from: https://doi.org/10.1038/nprot.2007.44
Komada M, Takao K, Miyakawa T. Elevated plus maze for mice. J Vis Exp. 2008 Dec;(22).
National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. (2011) 8th edition. Washington (DC): National Academies Press (US). Available from: https://www.ncbi.nlm.nih.gov/books/NBK54050/ doi: 10.17226/12910
Effect P, Supplement D, Induced C, Stress O, Models AR. Effect of Trévo Dietary Supplement on Caffeine Induced Oxidative Stress in Albino Rat Models. 2016;3(2):92–7.
Sharma D, Sangha GK. Triazophos induced oxidative stress and histomorphological changes in liver and kidney of female albino rats. Pestic Biochem Physiol. 2014; 110:71–80.
Reis R, Charehsaz M, Sipahi H, Ekici AID, Macit Ç, Akkaya H, et al. Energy Drink Induced Lipid Peroxidation and Oxidative Damage in Rat Liver and Brain When Used Alone or Combined with Alcohol. J Food Sci. 2017;82(4):1037–43.
Shirwaikar A, Rajendran K, Dinesh Kumar C, Bodla R. Antidiabetic activity of aqueous leaf extract of Annona squamosa in streptozotocin-nicotinamide type 2 diabetic rats. J Ethnopharmacol. 2004;91(1):171–5.
Worthley MI, Prabhu A, De Sciscio P, Schultz C, Sanders P, Willoughby SR. Detrimental effects of energy drink consumption on platelet and endothelial function. Am J Med. 2010;123(2):184–7.
Grasser EK, Yepuri G, Dulloo AG, Montani JP. Cardio- and cerebrovascular responses to the energy drink Red Bull in young adults: a randomized cross-over study. Eur J Nutr. 2014;53(7):1561–71.
Richards G, Smith AP. A Review of Energy Drinks and Mental Health, with a Focus on Stress, Anxiety, and Depression. J Caffeine Res. 2016 Jun;6(2):49–63.
Korte SM, De Boer SF. A robust animal model of state anxiety: fear-potentiated behaviour in the elevated plus-maze. Eur J Pharmacol. 2003;463(1–3):163–75.
Rodgers RJ, Dalvi A. Anxiety, defence and the elevated plus-maze. Neurosci Biobehav Rev. 1997 Nov;21(6):801–10.
Nehlig A, Daval JL, Debry G. Caffeine and the central nervous system: mechanisms of action, biochemical, metabolic and psychostimulant effects. Brain Res Brain Res Rev. 1992;17(2):139–70.
McLellan TM, Lieberman HR. Do energy drinks contain active components other than caffeine? Nutr Rev. 2012 Dec;70(12):730–44.
Bruce M, Scott N, Lader M, Marks V. The psychopharmacological and electrophysiological effects of single doses of caffeine in healthy human subjects. Br J Clin Pharmacol. 1986 Jul;22(1):81–7.
Klevebrant L, Frick A. Effects of caffeine on anxiety and panic attacks in patients with panic disorder: A systematic review and meta-analysis. Gen Hosp Psychiatry. 2022; 74:22–31.
El Yacoubi M, Ledent C, Parmentier M, Costentin J, Vaugeois JM. The anxiogenic-like effect of caffeine in two experimental procedures measuring anxiety in the mouse is not shared by selective A(2A) adenosine receptor antagonists. Psychopharmacology (Berl). 2000 Feb;148(2):153–63.
Bhattacharya SK, Satyan KS, Chakrabarti A. Anxiogenic action of caffeine: an experimental study in rats. J Psychopharmacol. 1997;11(3):219–24.
Al-Shaar L, Vercammen K, Lu C, Richardson S, Tamez M, Mattei J. Health Effects and Public Health Concerns of Energy Drink Consumption in the United States: A Mini-Review. Front Public Heal [Internet]. 2017;5. Available from: https://www.frontiersin.org/articles/10.3389/fpubh.2017.00225
Zeidán-Chuliá F, Gelain DP, Kolling EA, Rybarczyk-Filho JL, Ambrosi P, Terra SR, et al. Major components of energy drinks (caffeine, taurine, and guarana) exert cytotoxic effects on human neuronal SH-SY5Y cells by decreasing reactive oxygen species production. Oxid Med Cell Longev. 2013; 2013:791795.
Blaise JH, Park JE, Bellas NJ, Gitchell TM, Phan V. Caffeine consumption disrupts hippocampal long-term potentiation in freely behaving rats. Physiol Rep. 2018;6(5):e13632.
Published
Issue
Section
License
Copyright (c) 2023 Journal and Publisher

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
The Journal is owned, published and copyrighted by the Nigerian Medical Association, River state Branch. The copyright of papers published are vested in the journal and the publisher. In line with our open access policy and the Creative Commons Attribution License policy authors are allowed to share their work with an acknowledgement of the work's authorship and initial publication in this journal.
This is an open access journal which means that all content is freely available without charge to the user or his/her institution. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles in this journal without asking prior permission from the publisher or the author.
The use of general descriptive names, trade names, trademarks, and so forth in this publication, even if not specifically identified, does not imply that these names are not protected by the relevant laws and regulations. While the advice and information in this journal are believed to be true and accurate on the date of its going to press, neither the authors, the editors, nor the publisher can accept any legal responsibility for any errors or omissions that may be made. The publisher makes no warranty, express or implied, with respect to the material contained herein.
TNHJ also supports open access archiving of articles published in the journal after three months of publication. Authors are permitted and encouraged to post their work online (e.g, in institutional repositories or on their website) within the stated period, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access). All requests for permission for open access archiving outside this period should be sent to the editor via email to editor@tnhjph.com.














