Nigella sativa oil (NSO) as an adjuvant in the management of mild COVID-19 infection in Kaduna state
Main Article Content
Abstract
Background: To assess the efficacy of Nigella sativa oil (NSO) in the management of mild COVID-19 infection in Kaduna state.
Method: Quasi-experimental study among 51 mild COVID-19 cases enrolled in Hamdala isolation center from 27th October, 2020 to 20th May, 2021. Outcome variables were viral clearance, resolution of symptoms and duration of hospital stay after commencement of the different treatment regimen at level of significance P < 0.05 and effect size (Cohen’s D 0.2= small, 0.5=medium and ≥0.8 = large).
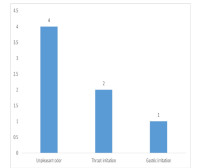
Result: Out of the 51 people enrolled in the study, 26 (51%) were placed on NSO plus usual care while 25 (49%) were on usual care alone with Mean age (SD) of 30.77±14.56 and 32.60±17.50 respectively. There were 16 (61.5%) females and 10 (38.5%) males in the NSO group and 19 (76%) females with 6 (24%) males in the usual care group. More patients on NSO have symptoms 12 (46.2%); ranging from fever, malaise, anosmia and loss of taste compared to 8 (32.0%) of the usual care group. Mean recovery time was significantly shorter 4.50±1.51 days in the NSO group, compared to 7.38±2.20 in the usual care with medium effect size (t-value = -3.483, Cohen’s D = 0.7, P = 0.003). Repeat PCR test was significantly different 48hours after commencement of treatment between groups, with large effect size (t=2.706, Cohen’s D=0.8, p=0.009).
Conclusion: NSO as add-on therapeutic agent was associated with faster recovery, viral clearance and shorter duration of care than usual care alone in patients with mild COVID-19 infection.
Downloads
Article Details
Section

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
The Journal is owned, published and copyrighted by the Nigerian Medical Association, River state Branch. The copyright of papers published are vested in the journal and the publisher. In line with our open access policy and the Creative Commons Attribution License policy authors are allowed to share their work with an acknowledgement of the work's authorship and initial publication in this journal.
This is an open access journal which means that all content is freely available without charge to the user or his/her institution. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles in this journal without asking prior permission from the publisher or the author.
The use of general descriptive names, trade names, trademarks, and so forth in this publication, even if not specifically identified, does not imply that these names are not protected by the relevant laws and regulations. While the advice and information in this journal are believed to be true and accurate on the date of its going to press, neither the authors, the editors, nor the publisher can accept any legal responsibility for any errors or omissions that may be made. The publisher makes no warranty, express or implied, with respect to the material contained herein.
TNHJ also supports open access archiving of articles published in the journal after three months of publication. Authors are permitted and encouraged to post their work online (e.g, in institutional repositories or on their website) within the stated period, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See The Effect of Open Access). All requests for permission for open access archiving outside this period should be sent to the editor via email to editor@tnhjph.com.
How to Cite
References
COVID NN. outbreak in Nigeria situation report—Abuja: Nigeria centre for disease control. Available on https://ncdc. gov.ng/themes/common/files/sitreps/0daa083aeed110eddba8937c1f90a6d9. pdf. On 27th November. 2020.
Idris U, Oyefabi AM, Dalhat MM, Umar IM, Abdulmajid M, Gwamna J, Mahmud AS. Treatment Outcomes in COVID-19 patients with comorbidities in Kaduna state, Northwestern Nigeria. Nigerian Health Journal. 2023 Apr 20;23(1):517-23.
Oyefabi A, Musa S, Kambai H, Usman I, Gwamna J, Sheyin J, Ige O, Abdullahi M, Sunday J, Kera HN, Atiku A. Comparison of the Ivermectin and Lopinavir/Ritonavir Treatment Outcomes among COVID-19 Mild to Moderate Cases in Kaduna State. West African Journal of Medicine. 2022 Feb 1;39(2):140-6.
Cao M, Zhang D, Wang Y, Lu Y, Zhu X, Li Y, Xue H, Lin Y, Zhang M, Sun Y, Yang Z. Clinical features of patients infected with the 2019 novel coronavirus (COVID-19) in Shanghai, China. MedRxiv. 2020 Mar 6:2020-03.
He J, Guo Y, Mao R, Zhang J. Proportion of asymptomatic coronavirus disease 2019: A systematic review and meta‐analysis. Journal of medical virology. 2021 Feb;93(2):820-30.
Idris U, Moses OA, Muhammad IU, Arinola OM, Muazu AS, Saulawa MA, Isah FM, Kera HN, Dalhat MM, Manga HB. Socio-Demographic and Clinical Characteristics and Outcomes of COVID-19 Patients in Kaduna State. Journal of Biochemistry, Microbiology and Biotechnology. 2023 Jul 31;11(1):7-10).
Stawicki SP, Jeanmonod R, Miller AC, Paladino L, Gaieski DF, Yaffee AQ, De Wulf A, Grover J, Papadimos TJ, Bloem C, Galwankar SC. The 2019–2020 novel coronavirus (severe acute respiratory syndrome coronavirus 2) pandemic: A joint american college of academic international medicine-world academic council of emergency medicine multidisciplinary COVID-19 working group consensus paper. Journal of global infectious diseases. 2020 Apr;12(2):47.
Rodriguez-Guerra M, Jadhav P, Vittorio TJ. Current treatment in COVID-19 disease: a rapid review. Drugs in Context. 2021;10.
Lin LT, Hsu WC, Lin CC. Antiviral natural products and herbal medicines. Journal of traditional and complementary medicine. 2014 Jan 1;4(1):24-35.
Shirvani H, Rostamkhani F, Arabzadeh E, Mohammadi F, Mohammadi F. Potential role of Nigella sativa supplementation with physical activity in prophylaxis and treatment of COVID-19: a contemporary review. Sport Sciences for Health. 2021 Dec;17(4):849-54.
Abel-Salam BK. Immunomodulatory effects of black seeds and garlic on alloxan-induced diabetes in albino rat. Allergologia et immunopathologia. 2012 Nov 1;40(6):336-40.
Abdel-Sater KA. Gastroprotective effects of Nigella Sativa oil on the formation of stress gastritis in hypothyroidal rats. International Journal of Physiology, Pathophysiology and Pharmacology. 2009;1(2):143.
Boskabady MH, Mohsenpoor N, Takaloo L. Antiasthmatic effect of Nigella sativa in airways of asthmatic patients. Phytomedicine. 2010 Aug 1;17(10):707-13.
Singh M, de Wit E. Antiviral agents for the treatment of COVID-19: Progress and challenges. Cell Reports Medicine. 2022 Mar 15;3(3).
Ahmad I. The race to treat COVID-19: Potential therapeutic agents for the prevention and treatment of SARS-CoV-2. European journal of medicinal chemistry. 2021 Mar 5; 213:113157.
Osibogun A, Balogun M, Abayomi A, Idris J, Kuyinu Y, Odukoya O, Wright O, Adeseun R, Mutiu B, Saka B, Osa N. Outcomes of COVID-19 patients with comorbidities in southwest Nigeria. PloS one. 2021 Mar 15;16(3):e0248281.
Sampaio Rocha-Filho PA, Albuquerque PM, Carvalho LC, Dandara Pereira Gama M, Magalhães JE. Headache, anosmia, ageusia and other neurological symptoms in COVID-19: a cross-sectional study. The Journal of Headache and Pain. 2022 Dec;23(1):1-1.
Koshak AE, Koshak EA, Mobeireek AF, Badawi MA, Wali SO, Malibary HM, Atwah AF, Alhamdan MM, Almalki RA, Madani TA. Nigella sativa for the treatment of COVID-19: An open-label randomized controlled clinical trial. Complementary Therapies in Medicine. 2021 Sep 1; 61:102769.
Ashraf S, Ashraf S, Ashraf M, Imran MA, Kalsoom L, Siddiqui UN, Ghufran M, Majeed N, Farooq I, Habib Z, Hilal A. Therapeutic efficacy of Honey and Nigella sativa against COVID-19: A multi-center randomized controlled clinical trial (HNS-COVID-PK). medRxiv. 2020 Jan 1.
Imran M, Khan SA, Abida, Alshammari MK, Alkhaldi SM, Alshammari FN, Kamal M, Alam O, Asdaq SM, Alzahrani AK, Jomah S. Nigella sativa L. and COVID-19: A glance at the anti-COVID-19 chemical constituents, clinical trials, inventions, and patent literature. Molecules. 2022 Apr 25;27(9):2750.
Tavakkoli A, Mahdian V, Razavi BM, Hosseinzadeh H. Review on clinical trials of black seed (Nigella sativa) and its active constituent, thymoquinone. Journal of pharmacopuncture. 2017 Sep;20(3):179.
Eğilmez OK, Kökten N, Kalcıoğlu MT, Ekici AI, Şerifler S, Yeşilada E. Investigation of the protective effect of Nigella sativa oil in cisplatin induced oral mucositis: an experimental study. Turkish archives of otorhinolaryngology. 2020 Mar;58(1):10.